 
For example, spiritual genograms provide a way to map significant events, affiliations, and family conflicts related to religion (Frame, 2000 Hodge, 2001). Given these factors and the overall flexibility of its use, the genogram is well-suited for generalist and clinical social work practice.Īlthough most genograms graphically represent multiple family generations and the quality of family relationships, genograms can be designed to capture other information, as well. Further, the experience of constructing a genogram with clients can be therapeutic in and of itself (Papadopoulos & Bor, 1997). 
The genogram can be useful in charting the basic family structure, recording individualized information (e.g., biological or legal details) to better understand relationship dynamics, and delineating family relationships (Connolly, 2005). As the genogram is helpful in understanding patterns and connections, it has been noted as “the supreme integrative tool” (Gerson, 1995, p. Seeing family behavior and relationships graphically can also help to reframe and normalize clients’ perspectives on their concerns. Identifying these connections can be useful when clients are stuck in a narrow view of the problem, blaming a particular family member for the issues of concern in the family. Genograms used with families may also be helpful in overcoming resistance “as they begin to see the connections between their concerns and historical family patterns” (McGoldrick, Gerson, & Petry, 2008, p. Similarly, the genogram invites the client to share personal identification in terms of race, gender, ethnicity, and cultural affiliation, which aligns with the discipline’s emphasis on understanding the various aspects of identity. For example, the genogram offers the opportunity to define and explore the family by acknowledging the client as the “expert.” This practice accounts for the evolving conceptualization of the family continually influenced by shifting cultural norms (Connolly, 2005). 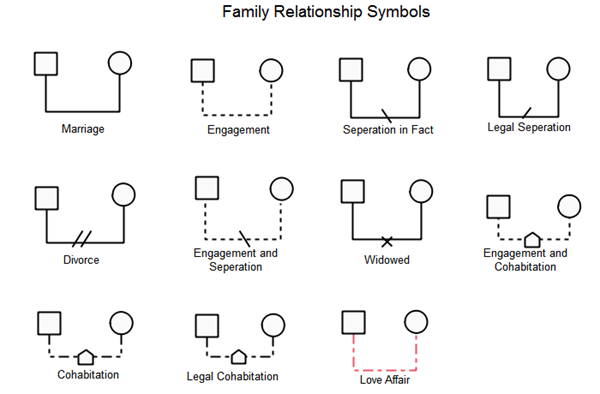
The genogram offers insight into that very understanding. Such a framework can help practitioners determine the sources of presenting issue(s) and the foci of interventions.Īdditionally, social work emphasizes the “not knowing” stance, acknowledging the client’s socially constructed understanding of his or her world and fit within that world. A central benefit is that this instrument introduces a client to the principles of systems theory, which are fundamental to social work practice (McGoldrick, Gerson, & Petry, 2008). The use of the genogram in social work practice is supported by the profession’s knowledge base as well as its values and ethical standards. In general, the genogram is useful in gathering information, understanding relationship dynamics and behavioral patterns, promoting the client’s self-understanding, conducting assessments, and guiding the practitioner to interventions. Typically, these are familial relationships. The genogram may be defined as a visual tool for exploring a client’s social relationships across time. Throughout this article, “client” will be used, but it could be interchangeable with client system when appropriate. It is noteworthy that the genogram can be useful for an individual client or a client system (i.e., a family). This article will discuss the use of genograms in social work and describe how to create a genogram using Microsoft Word. However, genograms can also be helpful when working with adults and serve as a tool to examine issues of interest beyond family dynamics. In child welfare practice, for example, the genogram is useful to incorporate changes over time, which is particularly helpful in charting shifts in custodial care for instances such as adoption or foster placements (Altshuler, 1999). Historically, the genogram is most commonly thought of in relation to practice with children, adolescents, and families to explore the quality of relationships and behavioral patterns across generations. Genograms are a practical tool in social work practice, both in terms of assessment and intervention. Depending on which version of Word you are using, instructions may vary slightly from those provided.) Also, please note that versions of software vary. (Editor's Note: To read this article with all illustrations, please download the Spring 2015 issue from the "digital issues" page of the magazine section of the site. Pope, Ph.D., LCSW, and Jacquelyn Lee, Ph.D., LCSW 
0 Comments
 Once the unicorn is colored, it should be cut out and glued to the left or right side of the wreath. This can then be colored in any way the child desires, and embellished with bows, pom-poms, rainbow stickers, stars, or puffy paint. 
This craft is easy for any child, and begins with cutting out a “wreath” from ordinary poster paper. Large craft sticks can be attached to the back for a great gift for mom or dad, or any pair the kids had in mind when creating their “characters.” 6. Once the unicorns are finished, they should be glued to thin cardboard and sealed in glassine. Yarn can be used for hair, red puffy paint for lipstick, and felt for a mustache or beard. The youngster should color two unicorns and decorate their faces to depict people. This fun craft can be a gift for mom and dad–or anyone else the child has in mind. Long strips of bright construction paper can then be glued to the bottom to create a rainbow colored fringe.ĭecorate the horn with gold glitter and add wiggle eyes to make this cute craft lifelike. Once the face is colored and cut out, it should be glued to a square background in any color the child prefers. This engaging craft is a great individual or classroom activity and a coloring page featuring a unicorn face is ideal. Punch a hole at the top for a piece of yarn or string, and embellish it with glitter or puffy paint for an adorable piece of wall art. Slide it behind one or two of the unicorn’s legs and glue it fast. Once it’s colored and cut from the page, cut a large, crescent shape from yellow or white poster board. To make super fun wall art, start with a unicorn page featuring a full-length unicorn. The unicorn horn should be reinforced with a toothpick, on which rubber bands or paperclips can be hung, and markers, crayons, pencils, or similar items can be placed inside. 
Next, the unicorn’s head and mane should be cut from the page after it’s colored, and glued to the front of the box with the unicorn horn sticking up past the rim.įour pom-poms can be used on the bottom four corners as “feet.” A Cute Place to Store Crayonsīegin this craft with an ordinary square cardboard box about four inches wide and four inches high. 
Using a small square of colorful paper, have each child write his or her name and attach it to the unicorn for an adorable table placeholder. When the unicorn is colored and cut out, it should be wrapped around the box.Ī colorful tail can be made from several strips of bright construction paper and the unicorn’s horn embellished with glitter or pom poms. A Happy Unicorn Placeholderįor a fun classroom activity, give each student an empty tissue box and a coloring page featuring a smiling unicorn. Here are 10 inexpensive, fun, and creative craft ideas you can do with your completed coloring page! 1. Unicorns are fascinating and fun, and all youngsters love to color them. 10 Craft Ideas to do With Unicorn Coloring Pages   He is survived by his wife of 50 years, the former Jean Crafton five children, a sister and a brother, according to CBS News. Osgood was also a writer, with six published books. He was inducted into the Radio Hall of Fame in 1990. 8, 1933, Osgood joined CBS News in September 1971 after four years with WCBS News Radio 88 in New York and previous stints with ABC News, WHCT and WGMS, according to the Radio Hall of Fame. It’s a paradox, but it’s true.”ĭuring the peak of his radio career, Osgood was reaching more than 11 million listeners each week, the Times reported.īorn in New York City on Jan. “You bring your own experience and emotions to radio that you don’t to television. “Television is wonderful and I don’t mean to take anything from it, but the fact is that a TV picture is very literally ‘what you see is what you get,’” he said in 1991, according to the newspaper. Portrait of journalist standing against green background. Female newscaster in suit, sitting in front of green screen, looking at camera.Greenscreen studio setup. Osgood continued to work for radio stations after leaving TV, where his sign-off was “See you on the radio,” The Los Angeles Times reported. Browse 810+ news anchor green screen stock photos and images available, or start a new search to explore more stock photos and images. 
“Osgood’s character put you at ease, while his wit and humanity drew you in whether reflecting on stories large or small.” Browse 1,574 authentic anchor background stock photos, high-res images, and pictures, or explore additional news anchor background or boat anchor background stock images to find the right photo at the right size and resolution for your project. “He had the understated ease of a Southern gentleman even though he came from New York and possessed perhaps one of the greatest voices to have ever graced network television,” Mauro said in a statement. At the time, then-NATAS president and CEO Bob Mauro remembered having crossed paths with Osgood before. In 2017, he was presented with the National Academy of Television Arts & Sciences’ News & Documentary Emmy Award for lifetime achievement. “Who wouldn’t want to be the one who gets to introduce these terrific storytellers and the producers and writers and others who put this wonderful show together?” he said. 
At the time of his retirement, a then-84-year-old Osgood said he continued working because “it’s been such a joy doing it!” according to CBS News. From 1994 until his retirement in 2016, Osgood anchored “CBS Sunday Mornings,” winning three Daytime Emmy awards for outstanding morning program.   
Thereby, a large number of buyers and a large market share are won, but at the expense of profitability. Instead of setting a high initial price to skim off each segment, market-penetration pricing refers to setting a low price for a new product to penetrate the market quickly and deeply. The opposite new product pricing strategy of price skimming is market-penetration pricing. Market-Penetration Pricing Market-Penetration Pricing – New Product Pricing And finally, competitors should not be in sight – if they are able to enter the market easily and undercut the high price, price-skimming does not work. Also, the costs of producing smaller must not be so high that they overshadow the advantage of charging more. The product’s quality and image must support the high initial price, and enough buyers must want the product at that price. Price-skimming makes sense only under certain conditions. However, this new product pricing strategy does not work in all cases. This way, the company skimmed off the maximum amount of revenue from the various segments of the market. Within a year, prices were dropped again. After this segment had been skimmed for six months, Apple dropped the price considerably to attract new buyers. The phones were, consequently, only purchased by customers who really wanted the new gadget and could afford to pay a high price for it. When it introduced the first iPhone, its initial price was rather high for a phone. An example for a company using this new product pricing strategy is Apple. Many companies inventing new products set high initial prices in order to skim revenues layer by layer from the market. As a result of this new product pricing strategy, the company makes fewer but more profitable sales. This means that the company lowers the price stepwise to skim maximum profit from each segment. Price-skimming (or market-skimming) calls for setting a high price for a new product to skim maximum revenues layer by layer from those segments willing to pay the high price. It is also referred to as market-skimming pricing. The first new product pricing strategies is called price-skimming. Let’s learn more about these two new product pricing strategies. Two new product pricing strategies are available: Price Skimming and Market Penetration Pricing. When companies bring out a new product, they face the challenge of setting prices for the very first time. One stage is particularly challenging: the introductory stage. Pricing strategies tend to change as a product goes through its product life cycle.   Compounds are classified as ionic or molecular (covalent) on the basis of the bonds present in them. Covalent bonds are the attractive forces between the positively charged nuclei of the bonded atoms and one or more pairs of electrons that are located between the atoms. When electrons are “shared” and molecules form, covalent bonds result. Ionic bonds are electrostatic forces of attraction, that is, the attractive forces experienced between objects of opposite electrical charge (in this case, cations and anions). When electrons are transferred and ions form, ionic bonds result. The nature of the attractive forces that hold atoms or ions together within a compound is the basis for classifying chemical bonding. Carbon will form an anion with a charge of 4−: C 4−, a carbide ion. Write the symbol for each ion and name them.Īl will form a cation with a charge of 3+: Al 3+, an aluminum ion. Predict which forms an anion, which forms a cation, and the charges of each ion. Check Your LearningĪluminum and carbon react to form an ionic compound. The symbol for the ion is N 3−, and it is called a nitride ion. Thus, a nitrogen atom will form an anion with three more electrons than protons and a charge of 3−. A nitrogen atom must gain three electrons to have the same number of electrons as an atom of the following noble gas, neon. Nitrogen’s position in the periodic table (group 15) reveals that it is a nonmetal. The symbol for the ion is Mg 2+, and it is called a magnesium ion. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. A magnesium atom must lose two electrons to have the same number electrons as an atom of the previous noble gas, neon. Magnesium’s position in the periodic table (group 2) tells us that it is a metal. For example, copper can form ions with a 1+ or 2+ charge, and iron can form ions with a 2+ or 3+ charge. In fact, transition metals and some other metals often exhibit variable charges that are not predictable by their location in the table. This trend can be used as a guide in many cases, but its predictive value decreases when moving toward the center of the periodic table. For example, group 17 elements (one group left of the noble gases) form 1− ions group 16 elements (two groups left) form 2− ions, and so on. Moving from the far right to the left on the periodic table, elements often form anions with a negative charge equal to the number of groups moved left from the noble gases. That is, group 1 elements form 1+ ions group 2 elements form 2+ ions, and so on. Moving from the far left to the right on the periodic table, main-group elements tend to form cations with a charge equal to the group number. Note the usefulness of the periodic table in predicting likely ion formation and charge (Figure 2). (A discussion of the theory supporting the favored status of noble gas electron numbers reflected in these predictive rules for ion formation is provided in a later chapter of this text.) It has the same number of electrons as atoms of the next noble gas, krypton, and is symbolized Br −. This results in an anion with 35 protons, 36 electrons, and a 1− charge. For example, the neutral bromine atom, with 35 protons and 35 electrons, can gain one electron to provide it with 36 electrons. 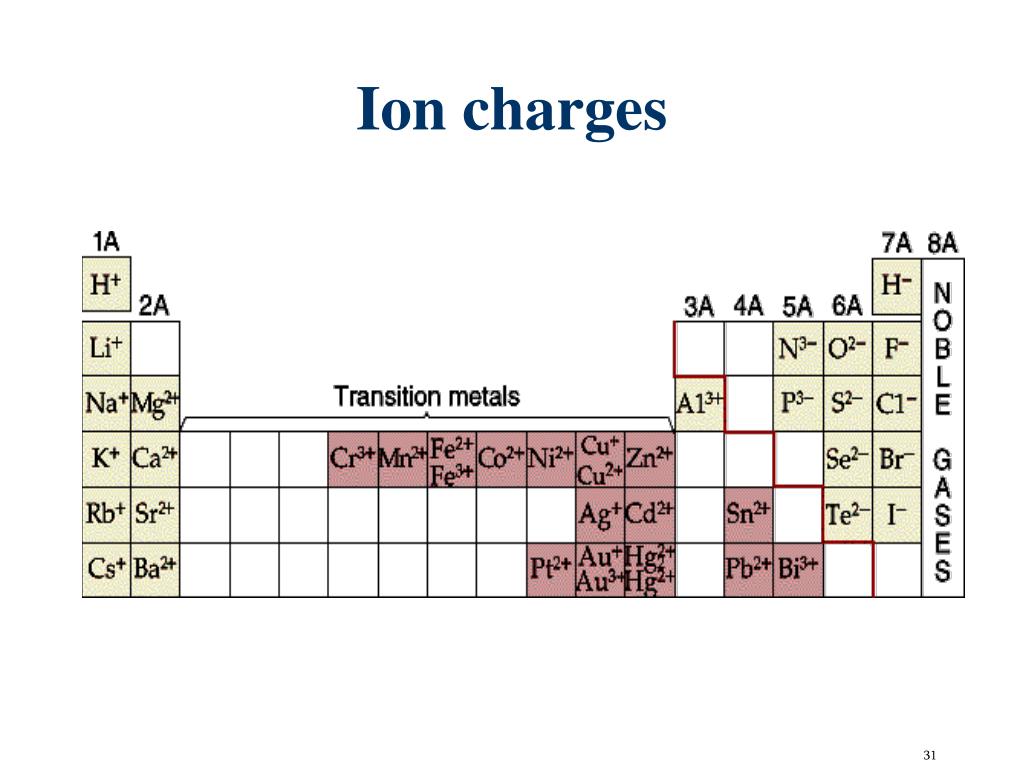
Atoms of group 17 gain one electron and form anions with a 1− charge atoms of group 16 gain two electrons and form ions with a 2− charge, and so on. When atoms of nonmetal elements form ions, they generally gain enough electrons to give them the same number of electrons as an atom of the next noble gas in the periodic table. The name of a metal ion is the same as the name of the metal atom from which it forms, so Ca 2+ is called a calcium ion. It has the same number of electrons as atoms of the preceding noble gas, argon, and is symbolized Ca 2+. 
This results in a cation with 20 protons, 18 electrons, and a 2+ charge. For example, a neutral calcium atom, with 20 protons and 20 electrons, readily loses two electrons. To illustrate, an atom of an alkali metal (group 1) loses one electron and forms a cation with a 1+ charge an alkaline earth metal (group 2) loses two electrons and forms a cation with a 2+ charge, and so on. Atoms of many main-group metals lose enough electrons to leave them with the same number of electrons as an atom of the preceding noble gas. You can use the periodic table to predict whether an atom will form an anion or a cation, and you can often predict the charge of the resulting ion. (b) A sodium cation (Na+) has lost an electron, so it has one more proton (11) than electrons (10), giving it an overall positive charge, signified by a superscripted plus sign. (a) A sodium atom (Na) has equal numbers of protons and electrons (11) and is uncharged.   
The critical angle is the point at which all of the incident light starts to totally reflect within the higher index medium. Whenever incident light travels from a high index medium to a low index medium (ie: glass to air), the critical angle is another useful tool for many optical applications including prisms. Special Case 2: Critical Angle (for Total Internal Reflection) To calculate the Brewster's Angle for a given material, use the following equation: 
Brewster's angle is often used in conjunction with polarizer coatings to minimize laser coupling loss and to maximize the signal strength of the desired light source. Brewster's angle has particular usefulness in optical applications where the P-Polarized signal (transmitting portion) must be optimized. For our simple air/glass interchange, Brewster's Angle is 56° 39′. Step 3: Once you have all four variables, use Fresnel's laws to calculate the loss in intensity due to reflection:Ī) Reflection loss for any angle of incidence:ī) Reflection loss for normal (0 degree) incidence:įor our air/glass interchange with normal incidence, reflection loss is about 4%.įor any given material, Brewster's Angle is the angle of incidence at which point the P-Polarization of the reflected portion, RP, is equal to 0. Step 2: Use Snell's law to define the relationship between n 1, n 2, θ 1 and θ 2 Θ 1 = angle of incidence (or incoming angle) = angle of reflection 
In the special case of a vacuum or air, n=1. However, it should be noted that the index of refraction for a given material is a function of wavelength and index increases as the wavelength gets shorter. All optical materials have a unique index of refraction that is a key characteristic of the material (ie: ZnSe, n=2.4). To Determine the answers to these questions, follow the steps below: Step 1: Define the variablesįind the index of refraction for both lower and higher mediums. What is the effect of polarization on the reflected and refracted portions of the incident light? What is the reflection loss of the incident light?ģ. What is the angle of refraction (θ 2) relative to the incident (θ 1) light? (It is generally known that the angle of reflection is equal to the angle of incidence).Ģ. There are three important considerations whenever this happens:ġ. Whenever an incident light traveling through one medium (ie: air with an index of refraction, denoted by "n", of 1) comes into contact with another medium with a high index of refraction (ie: glass with n=1.52), there is a reflected portion (bounces off the surface), and a refracted portion (transmits through the surface). We will also discuss two special cases, Brewster's Angle and the Critical Angle, which hae valuable applications in optics. In this section, we will discuss some of the basic behaviors of light when it travels from one medium to another. Tech Notes » Reflection & Refraction of Light Tutorial  |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
